Calcium carbonate
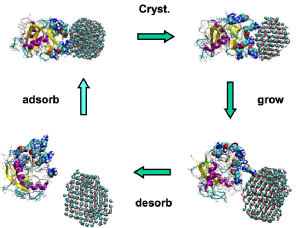
Calcium carbonate is a common biomineral found in shells, spicules and spines as well as other biological structures. Avian eggshells contain a layer of calcite. In chicken eggshells, it has been found that they also contain a large amount of the protein ovocleidin-17. Previous molecular dynamics simulations have shown that this protein acts as a catalyst in the nucleation of calcium carbonate as shown in the diagram below. The aim of this project was to investigate this interaction.
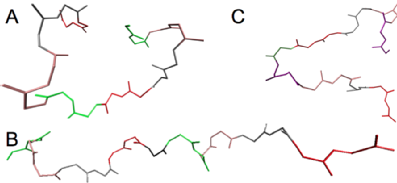
Three model peptides with doublets of charged residues were made, labelled A, B and C. The first two were chosen as they were similar to a protein found in geese eggshells. The introduction of a proline residue in the second peptide is expected to alter the secondary structure. The third peptide has lysine instead of arginine and a glycine residue in the middle. The glycine is expected to alter secondary structure of the peptide and the substitution with lysine will allow the effect of arginine on binding to be found.
Molecular dynamics simulations of these peptides showed the most energetically favoured conformation of each one which are shown below. The proline in the second peptide gave it the most sturdy structure. These conformations were used for further simulations.
Simulations of each peptide with an amorphous calcium carbonate nanoparticle were performed. In each case the peptide bound to the nanoparticle and changed conformation. By looking at which residues bound to the calcium carbonate nanoparticle in each case we can conclude that arginine is important for binding. The straight secondary structure of peptide B also seems to aid in the binding of the peptide to calcium carbonate.
I presented a poster on this project at the MOAC annual conference 2011 which can be downloaded by clicking on the icon below.
This project was supervised by Professor Mark Rodger
Reference: Freeman C.L.; Harding J.H.; Quigley D.; Rodger P.M. J. Phys. Chem. 2011, 115, 8175-8183
