Lignin degradation
Novel Enzymes for Lignin Degradation and Production of Renewable Chemicals
- to produce bio-ethanol via conversion to glucose and fermentation
- to produce chemical feedstocks for commercial use
However, a major obstacle to exploiting this renewable carbon is the durability of lignin, a heterogeneous aromatic polymer that encases the cellulose fibres in lignocellulose. Lignin, which comprises 10-30% of lignocellulose by dry weight, is composed or aryl-C3 units linked via a variety of C-C and C-O ether linkages that are extremely resistant to degradation.

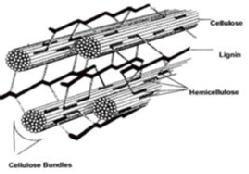
Current methods for bioethanol production utilise a chemical/physical pre-treatment step, in order to remove lignin and hemicellulose, and allow access to the cellulose content of the plant biomass. The most common method used for softwood pre-treatment is acid-catalysed steam pre-treatment (190-230 oC, 2-5% SO2). This is an energy-intensive process and reduces the carbon efficiency of biofuel production. If a biological process for lignin breakdown could be discovered, then it could be used to provide a biological pre-treatment step for 2ndgeneration bio-ethanol production, thereby increasing the bioavailability of cellulose, whilst requiring less energy input for pre-treatment. In addition, lignin degradation would release valuable low molecular weight aromatic chemicals that would add considerable value to biofuel production.
Study and Analysis of Lignin degradation
Lignin can be slowly degraded by white-rot fungi such as Phanerochaete chrysosporium, which produce an extracellular lignin peroxidase enzyme to commence the degradation process. Other fungal strains produce manganese peroxidase and laccase enzymes that are also active in lignin breakdown. However, in spite of a great deal of work on fungal lignin breakdown, fungal lignin-degrading enzymes have not been used for a commercially useful process for lignin breakdown.
 |
 |
Several types of bacteria are also known to degrade lignin, notably Actinomycetes such as Streptomyces viridosporus, and strains of Nocardia and Pseudomonas. The initial breakdown products are phenylpropanoid dimers which can be degraded by soil bacteria such as Sphingomonas and Pseudomonas via aromatic meta-cleavage pathways.
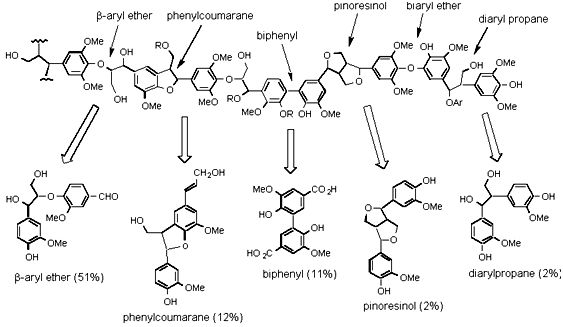
As part of the WOW (“Wealth out of Waste”) project, funded by IMRC, we are using novel spectrophotometric assays for lignin breakdown to identify new bacterial lignin degraders. We have developed two spectrophotometric assays to screen for lignin degradation activity.
The first assay involves the breakdown of fluorescently modified lignin, leading to an increase of fluorescence in solution. We have observed time-dependent increases in fluorescence over 0-2 hr, upon addition of cultures of known lignin-degrading Pseudomonas putida, Nocardia and Rhodococcus strains, relative to non-degrading strains. Similar levels of activity are observed using culture supernatant, in the presence of hydrogen peroxide, indicating that extracellular peroxidase enzymes are responsible, and activity is protein-dependent.
The second assay involves chemically nitrated lignin. Treatment of the nitrated lignin with bacterial lignin degraders leads to a time-dependent increase in absorbance at 400 nm, due to release of nitrophenol products. We are currently using these assays to screen for new lignin-degrading bacteria, and to examine the specificity of bacterial degraders towards different lignin types, and we are carrying out lignocellulose breakdown experiments, to examine the aromatic breakdown products.
