Fox Group
Synthetic and Medicinal Chemistry
We are interested in synthetic, mechanistic, medicinal and analytical organic chemistry in these areas:
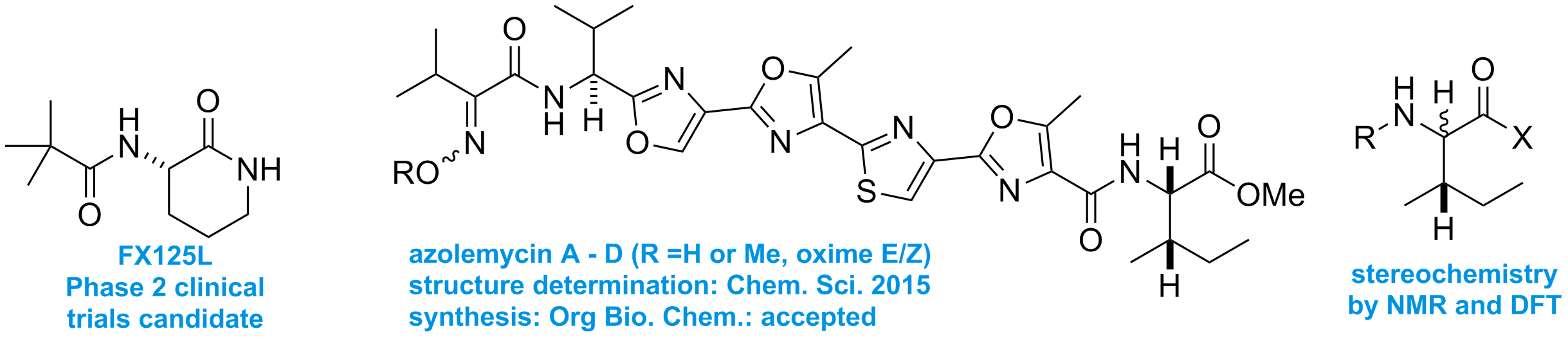
Drugs and other biologically active molecules. The medicinal chemistry for our anti-inflammatory acylamino-lactam project, started in Cambridge, ran in our lab until 2012. It involved the design and synthesis of new drug molecules in collaboration with Funxional Therapeutics Ltd. In 2012 the project was sold to Boehringer Ingelheim. We are continuing to work on the mechanism of action, and support further biological applications. We have designed new methods for the synthesis of unusual pharmacophores and are currently applying this to the synthesis of autophagy stimulators for the treatment of various diseases, in collaboration with E3Bio Ltd. We are also synthesising new anti-inflammatory compounds based on new asymmetric methods developed in our group, are looking to test these molecules in relevant functional biological assays. We are interested in developing cheap and scalable reagents and methods for the synthesis of unusual biologically active peptides, including natural products. We are interested in assigning stereochemistry (e.g isoleucine vs allo-isoleucine) via model system synthesis and DFT-based NMR spectrum calculation.
A video made by our 2021 URSSLink opens in a new window summer students about their syntheses of pyrazinones can be seen hereLink opens in a new window.
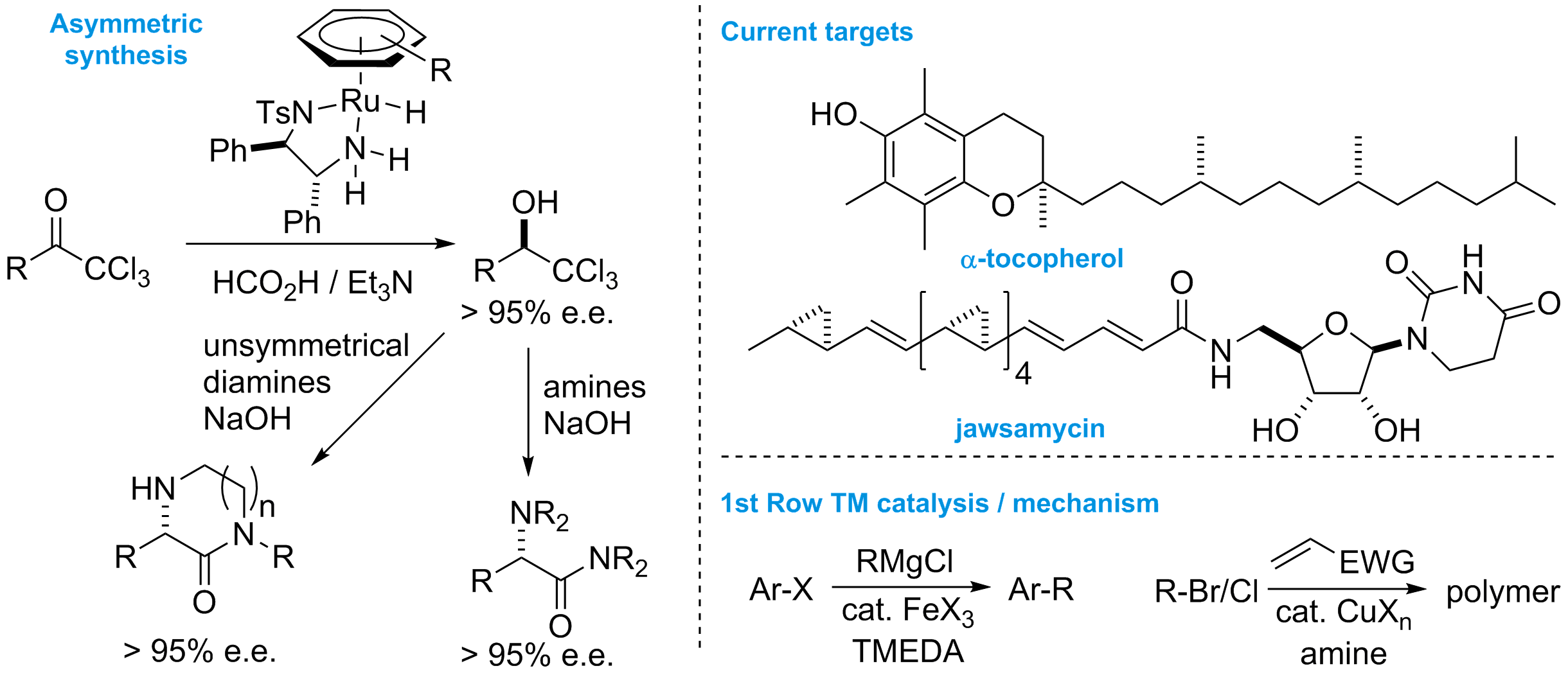
Catalysis in synthesis. Our on-going interest in reaction mechanism covers many aspects of synthesis, including transition-metal catalysis and organocatalysis for small molecule synthesis and polymerisation reactions. We analyse reactions with time-dependent and non-time-dependent kinetics analysis, DFT calculations and logical reasoning!
Asymmetric transfer hydrogenation of trichloromethyl-ketones produces enantiomerically-enriched trichloromethyl-alcohols. These molecules are extremely versatile chiral electrophiles that react with azide in Corey-Link reactions and, we have also shown, react with mono-amines and diamines to produce amino-amides, chiral heterocycles and 2-hetero-substituted carboxylic acids. We are currently developing these reactions further for the synthesis of natural products and new drug building blocks.
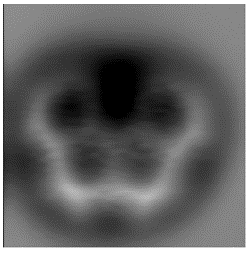
Molecular imaging as a new technique for organic synthesis - olympicene. The synthesis and imaging of benzo[cd]pyrene “olympicene” in collaboration with IBM, Zurich has led to the synthesis of a much wider series of polyaromatic hydrocarbons made from very simple starting materials in a single step. High resolution images obtained at IBM using Non-Contact AFM (NC-AFM) have identified new molecules in complex product mixtures and provided evidence for the elucidation reaction mechanisms. We are working on the isolation and imaging of other highly reactive, and otherwise unisolable, molecules, pushing the limits of the technique and analysing organic compounds that are usually only seen on paper.

 |
 |
 |
 |
Synthesis and characterisation of "non-Kekule" triangulene on a surface. Wanting to extend the "olympicene" idea, we made the precursor molecule dihydrotriangulene by "wet" chemistry methods and then our collaborators at IBM, Zurich removed two hydrogen atoms to produce the triplet ground state "diradical" triangulene for the first time and analysed it using non-contact AFM (NC-AFM) and scanning tunneling spectroscopy (STS) to see its structure and frontier orbitals. Link to paper is here.
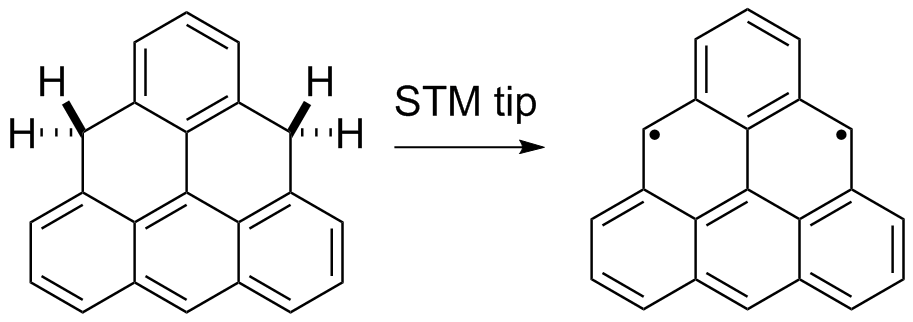
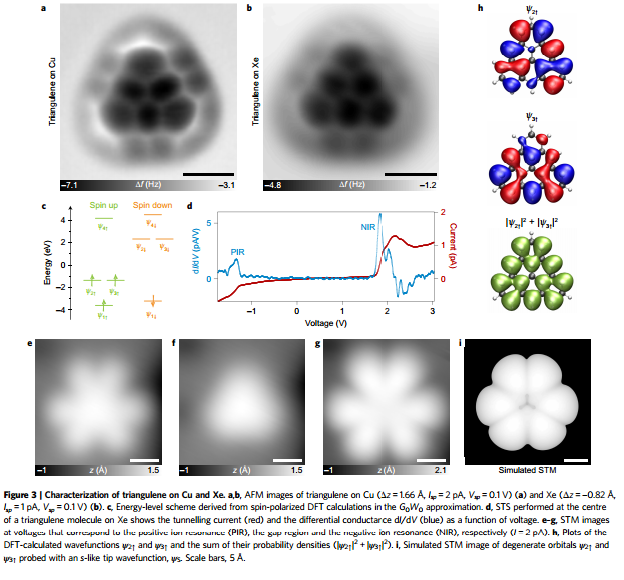

|
|
David Fox at the biotech blog DrugBaron on how to improve drug discovery hereLink opens in a new window. |
 |
David Fox at The Huffington Post on "olympicene" hereLink opens in a new window. |