John Blood
Contact Details
John Blood
MOAC DTC
Coventry House
University of Warwick
Gibbet Hill Road
Coventry
CV4 7AL
j.blood@warwick.ac.uk
Before MOAC
I was educated as a biochemist at Swansea University. During my time there I obtained a 2.1 and the faculty 'Reed Scientific Prize' for a outstanding report of my level 3 project on cyclising and purifying Stable Isotope Labelled cAMP from the corresponding AMP for use as an internal standard in Mass Spectrometry.
MSc Year
Since joining MOAC my scientific knowledge base has extended to biochemistry, chemistry, mathematics and computing. In addition to this my transferable skills have been vastly improved through a program of viva style examinations, giving presentations and producing posters.
1st Miniproject
My first eight week mini-project was the development of a mathematical model of erythrocyte carbohydrate metabolism with a view to altering Transketolase concentration to simulate administration of thiamine pyrophosphate. I used a series of ordinary differential equations to calculate the reaction velocity of each enzyme and subsequently updated the different pools of metabolites found in the glycolytic and pentose phosphate pathways. I achieved reasonable behaviour from a smaller pathway however the vast number of degrees of freedom associated with the huge number of molecules and enzymes associated with the two pathways lead to unpredictable behaviour. The following poster, presented at th Annual MOAC Conference 2009, shows the reactions I attempted to model (as well as a rather scary mug-shot of me).

2nd miniproject
My second and third mini project were linked, with the aim of investigating the structural basis of metal binding and dynamics in Metallothionein clusters. The second miniproject focused on the cloning and expression of plant metallothionies which are metal chelating proteins involved in the acquisition and transport of metal ions in plant cells. Domain 1 of the Ec metallothionine of four plants belonging to the pinophyta, containing a highly conserved metal-thiolate cluster motif, were to be cloned and expressed. Two methods of generating the EcI nucleotide insert, involving the use of PCR of a whole EcI/II construct and use of synthetic annealed primers were tested. Optimisation was attempted on the restriction digest of expression plasmid pET29b, production of insert by PCR and ligation of combined insert and vector. Transformation of the ligated pEcI construct into two sources of DH5α cells was attempted and various forms of construct confirmation were conducted including the use of restriction digest, PCR and DNA sequencing. Unfortunately none of the four constructs were generated in the eight week project, meaning only the previously isolated Triticum aestivum whole EcI/II could be used in the next miniproject.
3rd miniproject
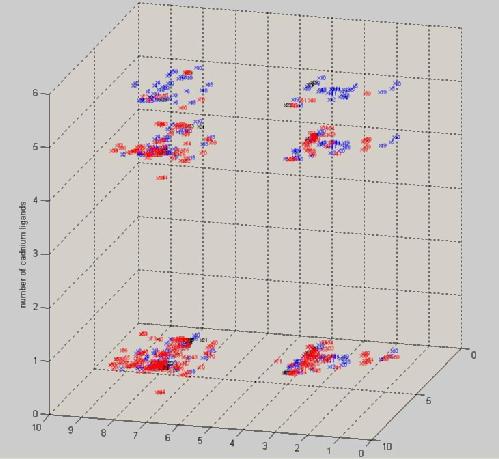
For my third miniproject I conducted Proton NMR studies of mixed Zn/Cd metalloforms EcI/II metallothionein. The main function of metallothioneins in vivo is sequestration of potentially toxic cadmium, copper and the essential cofactor zinc. Many metal atoms may be coordinated within the same protein due to the formation of metal thiolate clusters. Successful expression and purification of a metallothionine isolated from the plant Triticum aestivum was achieved. The ability of this EcI/II protein to coordinate a mixture of six Cd or Zn atoms, in combination with 2D [1H 1H] TOCSY and NOSEY nuclear magnetic resonance techniques were utilised to probe for structural and dynamical information. The ability of EcI/II to form two distinct forms of metal tholate cluster, M(II)2Cys6 and M(II)4Cys11His2, made it an excellent candidate for the study of metallothionine metal chelation. An increase in the cadmium content was shown to hasten the dynamics of the protein and analysis of 2D NMR peak data was conducted in an effort to characterise the relationship between metalloform and dynamics. The 3D plot below shows the TOCSY peak positions of domain 1 (blue), a linker region (black) and domain 2 (red) of EcI/II as the amount of Cadmium in the sample is increased. You may be able to see the peaks for domain 2 disappearing with increased cadmium binding corresponding to a more flexible conformation.
PhD Project
I am currently working on a PhD project entitled "Probing the Conformations and Dynamics of a Protein Folding Catalyst Using Single Molecule FRET Studies". My two supervisors for this project are Prof Robert Freedman at Life Sciences, University of Warwick and Dr David Clarke at Central Laser Facility, Rutherford Appleton Laboratory, Harwell Science and Innovation Campus. A good outline of the project is given in the poster below, which I presented at the Inter-DTC Conference 2010.



