3. Diamond Electrochemistry
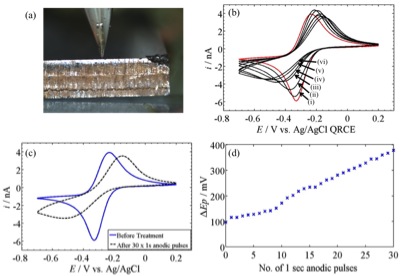
Diamond has a unique ability to be manipulated between stable H- (hydrophobic) and O- (hydrophilic) surface terminations. This study investigates how the electrochemical response of species is influenced by surface termination. In particular, the study investigates (i) how changing the surface termination on conducting (highly boron-doped >1020 B atoms cm-3) diamond electrodes influences the electrochemical response of inner-sphere species and (ii) how changing from H- to O- termination on semi-conducting (lowly boron-doped ~1018 B atoms cm-3) diamond inflences the electrochemical response of the outer-sphere mediator, Ru(NH3)63+, as shown in Fig. 1. These results have been applied towards a novel micropatterning technique of diamond electrode surfaces, as outlined in the publication below.
Publication: Patten, H. V., Hutton, L. A., Webb, J. R., Newton, M. E., Unwin, P. R., & Macpherson, J. V. (2015). Electrochemical “read–write” microscale patterning of boron doped diamond electrodes. Chemical Communications, 51(1), 164-167.