Oliver Sinfield
Current Work
I am currently studying for and PhD in Interdisciplinary Biomedical Research. My Research is conducted in the Lab of Prof. John McCarthy in the School of Life Sciences and Co-supervised with Prof. Robert Cross (Warwick Medical School). I am using microfluidics assisted fluorescence microscopy to study mRNP granules in the model eukaryotic system Saccharomyces cerevisiae, a budding yeast. You can read more about my PhD research here. I have worked as a demonstrator for undergraduates in their first year laboratory classes, teaching basic biochemistry and molecular techniques, there is a breif summary here. I have also set up a number of time saving and useful systems in our lab, including compiling a homemade Gibson Assembly kit and guide and sets of plasmids for protein and mRNA tagging and CRISPR Genome engineering in S. cerevisiae. My extra projects can be viewed here and plasmids are available on request.
My previous education
BSc (Hons) Biomedical Science - University of Warwick (2009-2012) (2:1)
For my undergraduate degree I studied Biomedical Science at the Universty of Warwick.
Undergraduate Project
My undergraduate project focused on using molecular genetics techniques to elucidate the conservation of identical duplicate genes in Saccharomyces cerevisiae. This project was based in the school of life sciences at the University of Warwick and was supervised by Profesor John McCarthy.
MSc Interdisciplinary Biomedical Research - Warwick Medical School (2012-2013) (Distinction)
I completed my MSc with distinction during the 2012-2013 academic year. This course involved 2 terms of taught classes and examinations followed by 2 12-week research projects in laboratories at the University of Warwick. My first project was in the Lab of Prof. John McCarthy (School of Life Sciences), where I am now studying for my PhD, in which I used fluorescence microscopy to study the role of eIF4G in the assembly of mRNP granules in S. cerevisiae. My second research project was carried out in the Lab of Dr. Steven Royle in the division of Biomedical Cell Biology, Warwick Medical School. In this project I used various imaging techniques such as fluorescent fusion protein labelling, immunofluorescence and 'Knock-sideways' to investigate a specific protein family linking membrane trafficking and mitosis. You can see breif summaries of my project below.
Miniproject 1 - Imaging the packaging of mRNA into stress granules.
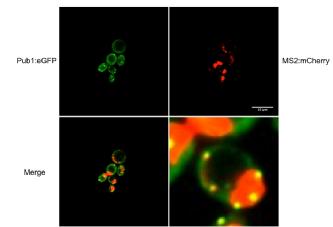
Stress Granules are thought to be sites of storage for translational pre-initiation complexes during stress conditions in Saccharomyces cerevisiae and are conserved in higher eukaryotes. DED1, a binding partner of eIF4G1 has been shown to affect the assembly of stress granules. To investigate this interaction further, plasmids and chromosomal integrations containing fluorescent marker proteins were used to tag known components of stress granules, mRNA and P bodies and study their interaction in yeast strains with deletions of the two eIF4G isoforms and overexpression plasmids of eIF4G2. eIF4G2 overexpression was found to cause significant increases in stress granule accumulation in eIF4G1 deletion background strains.
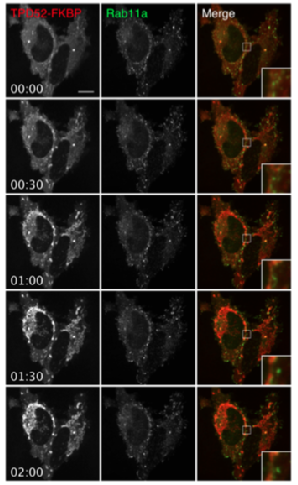 Mini-project 2 - Functional analysis of Tumour Protein D52-like proteins
Mini-project 2 - Functional analysis of Tumour Protein D52-like proteins
TPD52 like proteins are members of a family of small coiled-coil proteins, which have been shown to be specific targets of gene amplification in cancers. Studies in Acinar (polarized pancreatic secretory cells) have suggested a role in lysozomal membrane trafficking, and endocytosis to the apical membrane. Other studies have shown increased multinucleation in CHO-KI cell lines when TPD’s are overexpressed, as well the suspected presence of TPD52 at the cleavage furrow during cytokinesis, indicated by VAMP-8 positive vesicles found between nuclei. I used molecular biological techniques and microscopy to further elucidate the role of TPD52 like proteins TPD52 and TPD52L2, especially concentrating on their role in membrane trafficking and cytokinesis. The figure to the left shows a timelapse image of TPD52 relocalising to the mitochondria on addition of Rapamycin in HeLa cells, using a technique known as 'Knock-sideways'.

Oliver Sinfield
Contact Information
McCarthy Lab
School of Life Sciences
The University of Warwick
Coventry, CV4 7AL UK
Office: +44 (0) 2476574242
Lab: +44 (0) 2476522440
O dot Sinfield at warwick dot ac dot uk
