Biophysical measurements on FtsZ
Verification of FtsZ activity using light scattering.
Light scattering measurements are an excellent way of measuring whether FtzZ polymerises into protofilaments when GTP is added.
Figure 9 shows that the light scattering increases when GTP is added, plateaues when all the FtsZ is polymerised, and then drops off when the GTP is exhausted and no longer able to sustain the polymerisation. The red and orange curves show that it is possible to initiate a second round of polymerisation by adding more GTP.
This is all much as has been previously observed (Mukherjee and Lutkenhaus 1998) and were taken as confirming the presence of active FtsZ.
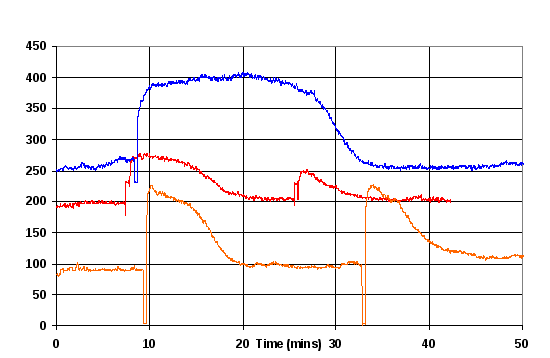
Figure 9 Monitoring FtsZ polymerisation using 90° light scattering of 100 μM polymerisation buffer containing 11 μM FtsZ and 50mM MES buffer. Blue: Batch A at pH 5.5. Red Batch A at pH 6.5. Orange Batch B at pH 6.5. Jumps in amplitude correspond to addition of GTP.
Batch B of the FtsZ produced a change in light scattering that was twice that of Batch A, even when the quantities used were adjusted to ensure the FtsZ was at 11 μM in both cases
Analytical Ultracentrifugation (AUC) analysis
The next test was to see if the FtsZ was monomeric, which was confirmed using AUC (Figure 10) and confirms that the FtsZ is predominantly monomeric. There are peaks that probably correspond to specific slightly more stable polymer lengths, such as the peak at 165 kDa which matches the molecular weight of an FtsZ tetramer.
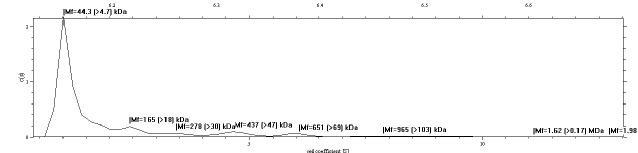
Figure 10 Approximation distribution of molecular mass of Batch A of FtsZ. The large peak at 44.3 kDa is the monomeric FtsZ (MW 40.3 kDa), with small quantities of longer polymers. The peak at 165 kDa suggests that there may be a stable tetramer configuration.
Abnormal light scattering measurements
While the very first light scattering measurements yielded very good results, there were some days when the light scattering results were consistently poor. Figure 11 is an example of some of the results that were obtained for FtsZ in polymerisation buffer before GTP was added. These results are very variable, and 10 times larger than those seen at other times. No problem was identified with the protocol, and removing and cleaning the outside of the cuvette made little difference.
No satisfactory explanation can be offered for these results, which are included in the thesis for the record, so that they can be referenced if similar problems reoccur.
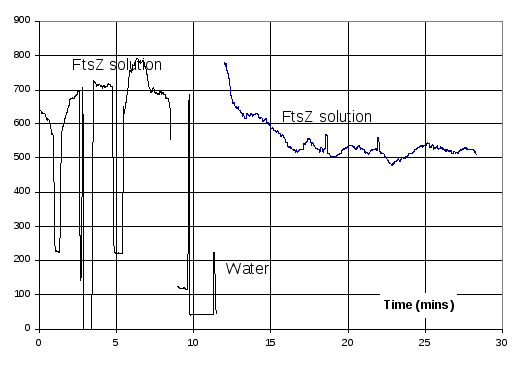
Figure 11 Example of abnormal light scattering results. Period of up to 8 minutes is of 11 μM FtsZ in polymerisation buffer. Values in the region 500-800 are light scattering results of the solution. Regions below 300 are when the cuvette was removed to be cleaned and inspected. At the 10 minute mark reference light scattering measurements were made on water to verify the operation of the fluorimeter, and then after 12 minutes the original FtsZ solution was replaced.
Control tests for linear dichroism measurements
Prior to using linear dichroism (LD) to measure the GTP initiated FtsZ protofilament formation, a control was performed without GTP where light scattering was used to monitor the mechanical effect on the FtsZ of the cuvette flow within the linear dichroism cell, by comparing light scattering results of the FtsZ in the polymerisation buffer before and after being subjected to Couette flow.
500 μL of Batch A FtsZ in polymerisation buffer was prepared and filtered. Light scattering was used to monitor GTP initiated protofilament formation in 100 μL FtsZ solution prepared using this buffer (Figure 12) to verify the integrity of the FtsZ. A second 200 μL batch of FtsZ solution was prepared and light scattering used to confirm its similarity to the first batch (Figure 12).
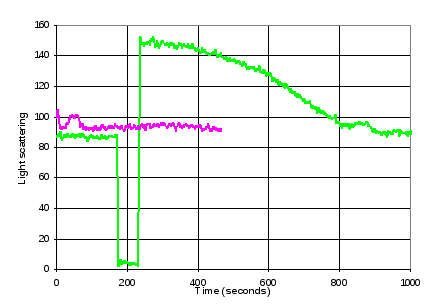
Figure 12. Verification of FtsZ activity. Green line: < 200 seconds: Light scattering of FtsZ in buffer solution. > 200 seconds: Increase and subsequent decay of light scattering as a result of addition of GTP. Purple line: Light scattering of second FtsZ solution, prior to LD measurements.
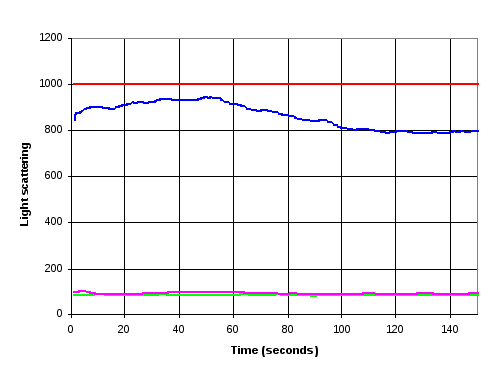
Figure 13 Comparison of light scattering results for FtsZ solution before (purple and green lines) and after (blue and red lines) LD measurements. Green and Purple lines as Figure 12. The red line indicates light scattering results after the FtsZ solution was used to perform two Linear Dichroism measurements using nested capillary tube. The blue line is after LD measurements using 2.5 mm quartz rod in capillary.
The LD of two samples of the solution was measured between 180 nm and 350 nm: the first using the nested capillary tube and the second using a conventional cell with a central quartz rod. Two measurements were made using the nested capillary tube, each an average of three runs, whilst only one measurement, consisting of an average of two runs, was made with the cell based on the quartz rod. In all cases, there was no discernable LD over the frequency range measured, as expected in that no ligands had been introduced to induce protofilament formation.
The samples were then extracted, and returned to the fluorimeter for further light scattering measurements (Figure 13). The sample from the nested capillary tube was slightly milky; an observation that was confirmed by a light scattering measurement that was 1000, indicating that it was larger than the detectors measurement range. It remained at this value for 15 minutes when measurements were stopped. The sample from the LD cell with the quartz rod gave a value of around 900, which drifted over time but showed no sign of reducing.
Investigation into light scattering variation due to mechanical agitation
The results in section 4.4 suggested that the FstZ solution might be sensitive to the mechanical stresses that are present when the fluid is sheared within the cuvette cell. This was of some concern in that sonication had been used to lyse the cells, and there was a possibility that the mechanical stresses associated with sonication might have had a detrimental effect at that stage.
Sonication was used as a standard way of measuring the sensitivity to mechanical stress. 100 μL of an 11 μM solution of FtsZ in polymerisation buffer was subjected to 2 minutes of sonication, and then protein conglomeration measured using light scattering. This was repeated with Batch A, B and RPG and in each case the light scattering value was 1000, the maximum value that can be recorded by the detector. This demonstrates that this characteristic is not specific to one of the FtsZ batches.
It appears to show a large protein aggregates with no significant sign of linear polymers. These are large enough to generate a signal when analysed using light scattering, but the lack of a filamentous structure would make them invisible to LD measurements.
The behaviour of the FtsZ solution under less severe mechanical stress was investigated by subjecting the FtsZ solution to 15 seconds of sonication prior to making measurements of GTP induced polymerisation. A suspicion that the effect may be linked to dissolved gases was also investigated by degassing some of the buffer solutions prior to testing. The results are shown in Figure 15.
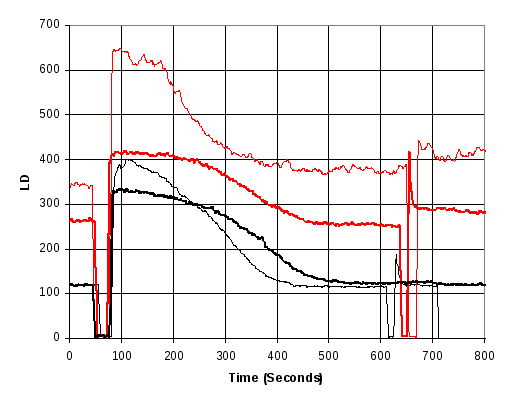
Figure 15 Increase in light scattering seen as a result of adding 0.2 mM GTP at approx 70 seconds and agitating by pipette stirring shortly after 600 seconds. Red curves indicate that the FtsZ in the polymerisation buffer had been sonicated for 15 seconds prior to starting the measurement. Heavy curves indicate that the sonication buffer had been degassed prior to adding GTP.
The results of Figure 15 show a number of features: First, removing the gas from the solution results in a longer, flatter steady state light scattering signal before it drops as a result of GTP depletion. Secondly: sonication induces an increase in the baseline light scattering signal, and appears to increase the measurement noise and thirdly, degassing the solution reduces the effect of the sonication. (8 provides further supporting data from batch A).
On the basis of this evidence all subsequent tests were performed with degassed solutions to reduce the variability that would arise from varying amounts of dissolved air, including the anomalous results that were believed to arise from air bubbles forming and steadily growing (8) and to reduce the susceptibility to mechanical stress.
FtsZ linear dichroism measurements
One of the goals of the mini-project was to use the new LD cell to perform kinetics measurements on FtsZ polymerisation. Before making dynamic measurements, the LD spectrum was measured of GTP polymerised FtsZ using the new cell and a conventional cell (Figure 16) to verify that LD signals from polymerised FtsZ could be seen using the nested capillaries.
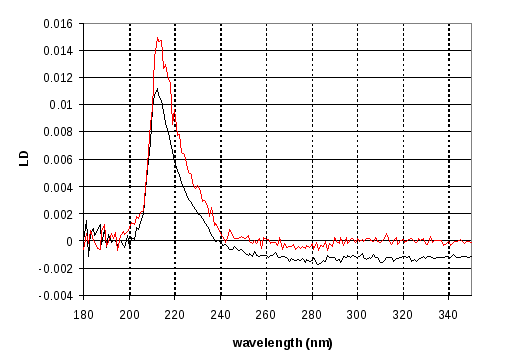
Figure 16 Linear Dichroism of GTP stabilised FtsZ protofilaments using a conventional LD cell (black) and the new nested capillary cell (red). The new cell results are slightly more noisy, and have a 212 nm peak (due to alpha helixes in the FTsZ and an indication of overall fibre alignment), which is slightly offset to higher wavelengths, although the peak value is 212 nm in both cases.
Figure 17 shows the results of a kinetics run using the nested capillary LD cell where the GTP was injected while the cell was not rotating, which will result in the GTP remaining in the base of the capillary, and then the cell rotation and LD measurements started immediately afterwards. The results show that the curve obtained is very close to results obtained using light scattering, suitably scaled and offset in time and amplitude.
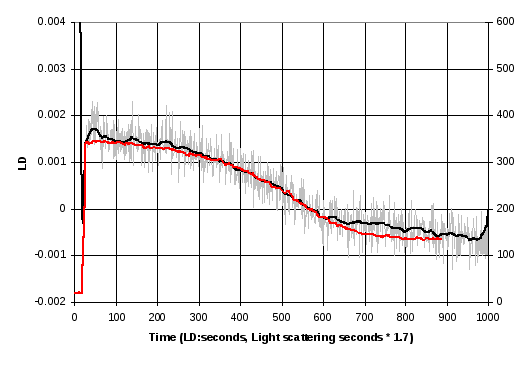
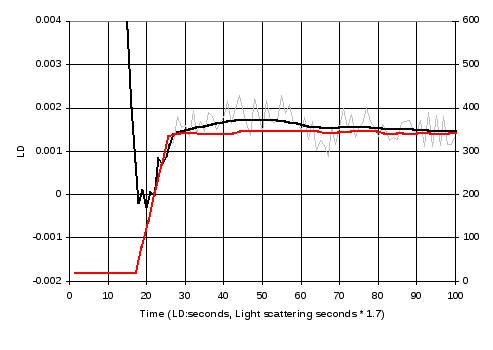
Figure 17 Kinetic measurement of LD signal at 212 nm (grey), and a smoothed version of these data after 25 seconds (black) and comparison with a curve obtained using light scattering. The LD and light scattering data were gathered from different experiments and the time scales of the light scattering results had to be multiplied by 1.7 for best alignment. The lower graph shows the same data, expanding the first 100 seconds.
A second run was performed (Figure 18) where the outer capillary was rotated and LD was measured as the GTP was injected. In this measurement, the LD signal was twice the amplitude of the previous meassurements, and there was a longer plateau. At the start, there was a relatively gradual increase in the measured LD extending over 60 seconds, followed by a large peak, after which the measured LD held constant for approximately 400 seconds before returning to the base level.
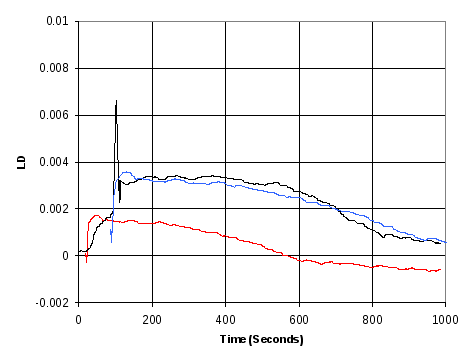
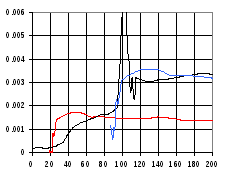
Figure 18 LD Kinetic measurement of FtsZ polymerisation (black) when GTP is injected while outer capillary is rotating, allowing LD measurements to be performed during this period. The results are compared with LD results from Figure 17 (red) and with these same results shifted and scaled to achieve best match of the curves (blue).
